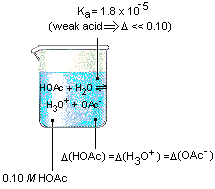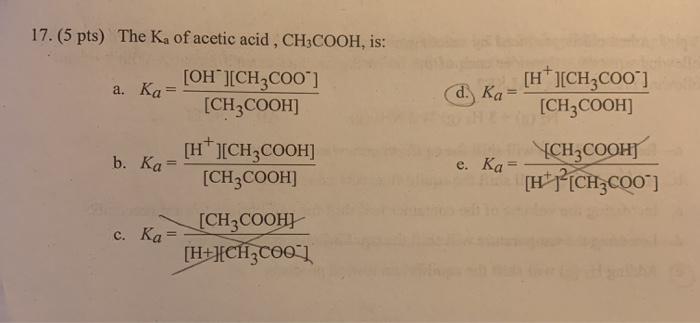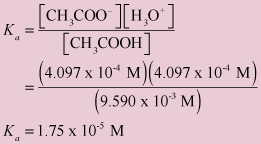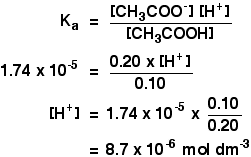![The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ] The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]](https://i.ytimg.com/vi/AufT6_CoFWY/maxresdefault.jpg)
The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]
THE DISSOCIATION CONSTANT OF ACETIC ACID FROM 0 TO 35° CENTIGRADE1 | Journal of the American Chemical Society
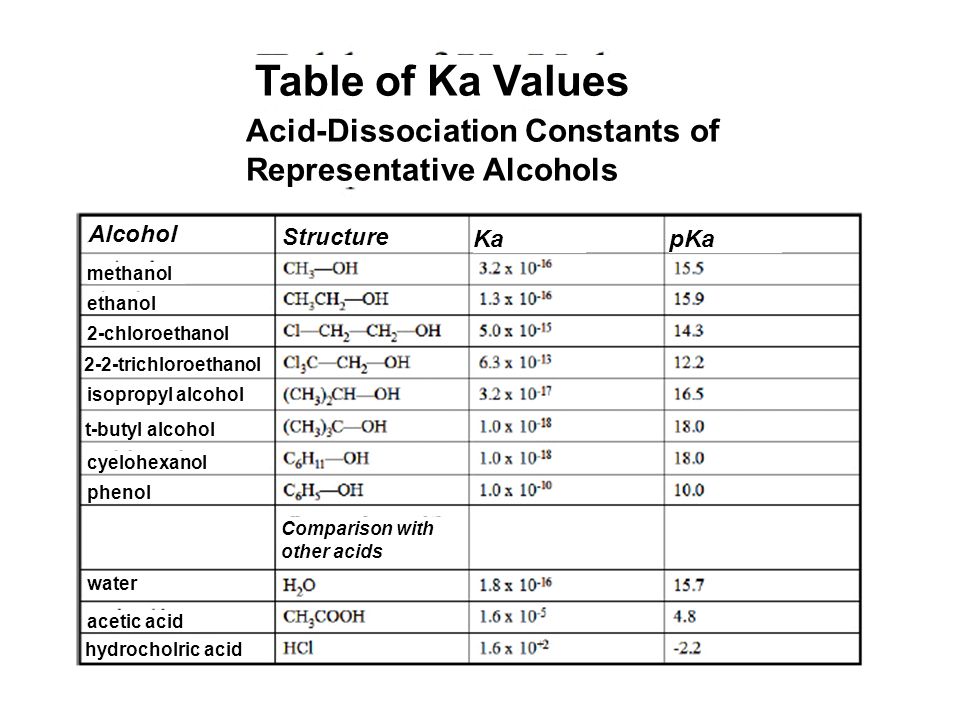
Table of Ka Values Acid-Dissociation Constants of Representative Alcohols Alcohol Structure Ka pKa methanol ethanol 2-chloroethanol 2-2-trichloroethanol. - ppt video online download
✓ Solved: Ka for acetic acid is 1.7× 10^-5 at 25°C. A buffer solution is made by mixing 52.1 mL of 0.122...

The Ka value for acetic acid, CH3COOH(aq), is 1.8x10^-5. Calculate the ph of a 2.80 M acetic acid solution - Home Work Help - Learn CBSE Forum
The pH of an acetic acid solution is 3.26. What is the concentration of acetic acid and what is the percent of acid that's ionized? - Quora

A student prepared 0.10M acetic acid solution and experimentally measured its pH to be 2.88. Calculate Ka - Brainly.in
![What is the pH of a 1 M CH3COOH solution? [ Ka of acetic acid = 1.8 × 10^-5, Kw = 10^-14 mol^2 litre^-2 ] What is the pH of a 1 M CH3COOH solution? [ Ka of acetic acid = 1.8 × 10^-5, Kw = 10^-14 mol^2 litre^-2 ]](https://i.ytimg.com/vi/5MXjDjLyUp4/maxresdefault.jpg)
What is the pH of a 1 M CH3COOH solution? [ Ka of acetic acid = 1.8 × 10^-5, Kw = 10^-14 mol^2 litre^-2 ]

How To Calculate the PH of a Buffer Solution | Equation & Example - Video & Lesson Transcript | Study.com